1. BCM 95® – Curcumin Formulated with Turmeric Oil
An example of this approach in market is BCM- 95®/BIOCURCUMAXTM, a “Curcuminoids Complex with Volatile compounds not less than 95%”. The product was patented in 2002 and the patent method discusses increasing the bioavailability of 98% pure Curcumin from a presumptive benchmark levels of 60-65% to 90-97% levels with added turmerone- enriched oil.
The presumption the marketers make in their bioavailability claim for Curcumin is based on the measurement of Curcumin in an unscientific approach due to the spectroscopic method used to estimate unabsorbed Curcumin in the fecal matter. Another anomaly in the study mentioned in the patent actually mentions range of absorption of 60-65% for unmodified Curcumin, which would be considered an extremely bioavailable compound by any standards, therefore this measurement in the bioavailability context, casts a long shadow of doubt on the protocol and analytical methodology adopted in the study which was conducted to prove the bioavailability of BCM-95® only.
Turmeric essential oil has not been proven to be safe for humans, and one study by Prof. Janet Funk clearly showed that Tumeric oil has a detrimental effect on the protective actions of Curcuminoids 18.
2. MERIVA® – CURCUMIN FORMULATED WITH PHOSPHATIDYLCHOLINE
A second formulation consisting of Curcumin (20%), Phosphatidylcholine (40%) and microcrystalline cellulose has been reported to increase the bioavailability of Curcuminoids 29 times 19.
However, what the product actually does is increase the concentration of the phase II metabolic products, namely the glucuronides and sulfates, which are the inactive forms of Curcumin. These glucuronides, as recounted earlier, neither display activity against cancer cells nor inhibit pro-inflammatory NFkB. Their bioavailability claim is based on a single human study involving only nine subjects divided in three arms of the study (three subjects each).
Studies on this brand of ‘enhanced’ Curcumin only substantiate the known anti-inflammatory effects of curcuminoids, but do not show or prove the benefits of any enhanced bioavailability on their existing results. The studies conducted only demonstrate the end points which were achieved by previous studies using Curcuminoids alone.
A new report on phosphatidylcholine appeared in the Nature News and Views article published in the columnCardiovascular Disease20 that reviewed the evidence high-fat foods are rich in the lipid phosphatidylcholine – the ingredient used in Meriva® – and its metabolite choline. Intestinal bacteria convert phosphatidylcholine and choline into Trimethylamine oxide (TMAO). The liver enzyme FMO3 converts Trimethylamine (TMA) into TMAO – a metabolite that is then found in the blood. Research conducted by Wang et al. as published in Nature21 showed that circulating levels of TMAO may contribute to greater plaque formation in the arteries, and could result in heart disease.
3. CURCUWIN™ – CURCUMIN FORMULATED WITH ULTRASOL™ TECHNOLOGY
The manufacturer of a product called CurcuWIN™ claims 46 times greater Curcumin bioavailability compared to standard Curcumin, BCM-95® or phytosomal Curcumin, Meriva®. The product claims to use an unknown ‘UltraSOLTM nutrient technology’, which increases all of the pharmacologically inactive Curcumin conjugates, including the glucuronides and sulfates.
4. THERACURMIN® – CURCUMIN USING NANOTECHNOLOGY
An ‘enhanced’ Curcumin product called Theracurmin® using a nanotechnology and water dispersion technology claims 40 times greater bioavailability in rats and 27 times higher bioavailability in humans compared to powdered Curcumin 22.
This technology also increases all of the pharmacologically inactive Curcumin conjugates, namely the glucuronides and sulfates.
5. LONGVIDA® – CURCUMIN FORMULATED INTO SOLID LIPID PARTICLES USING LECITHIN
Longvida® is marketed as “solid lipid particle formulation of curcumin”23. The manufacturer claims that their technology increases the bioavailability of Curcumin 65 times and states that their Curcumin detected in blood serum is free Curcumin and not glucuronidated or conjugated Curcumin metabolites. However no detailed toxicity studies are available on such products of recent origin. Here again lecithin could produce TMAO, a cardiotoxic chemical.
6. CAVAMAX® W8 – CURCUMIN FORMULATED WITH CYCLODEXTRIN
CAVAMAX® W8 is an ‘enhanced’ Curcumin made by complexing Curcumin with gamma cyclodextrin. The in vivobioavailability was measured in rats and showed a 10 to 20 times higher total curcuminoids. According to their research “Animals that received CAVAMAX® W8 curcumin showed a 10 to 20 times higher amount of total curcuminoids in their blood plasma, expressed as the sum of free curcumin and its metabolites (curcumin sulfates and curcumin glucuronides), than animals that received a commercial product or pure curcumin powder. This huge difference in HPLC- measured curcumin metabolites indicates that a maximum amount of curcumin was delivered into the blood stream of the rats, which can only be explained by the very highly bioavailable CAVAMAX® “W8 curcumin.”
In a human serum pharmacokinetic study, the manufacturer concluded that “Curcumin uptake was at least 4.4 times higher than the next-best commercial curcumin formulation in this clinical study.” The problem with these measurements in the rat and human study is that they estimate inactive metabolites, assuming that the metabolites are active, which they are not.
Cyclodextrin complexes are often used to increase the bioavailability of natural compounds. However, there is a toxicity issue where some cyclodextrins solubilize cholesterol, which can then be depleted from cell membranes. Beta cyclodextrin has a regulatory limit of 5 mg/kg/day because it is known to deplete cholesterol from cell membranes in this manner24.
Another toxicity issue has been revealed in rat studies where cyclodextrins were thought to increase the solubility and absorption of toxic contaminants in ingested food 25.
7. CURCUMIN FORMULATED WITH BIOPERINE®
This combination consist of two GRAS, patented ingredients Curcumin C3 Complex® and BioPerine®, a Piper nigrumextract. BioPerine® has been clinically studied for increasing the bioavailability of several nutrients including Curcuminoids. In a clinical study conducted in 1998 on healthy volunteers, it was found that BioPerine® can increase the bioavailability of curcuminoids by 20 folds26. This increase was measured for the Curcumin itself and not for its phase II metabolites. One of the reasons for this finding comes from the fact that the BioPerine® consists of piperine which is known to inhibit the formation of phase II metabolites because of glucuronidation. Piperine reduces glucuronidation by inhibition of UDP- glucuronyl transferase activity and by lowering the levels of endogenous glucuronic acid. Curcumin has tendency to undergo rapid biotransformation, its conversion in particular to Phase II metabolism is often seen as detrimental to bioactivity. BioPerine® helps in inhibiting the formation of phase II metabolites of Curcumin27. Piperine promotes nature’s own mechanism namely the flippase activity by P-gp inhibition and reduces the glucoronidation of Curcumin. This effect is short lived and requires simultaneous consumption of 5 mg of BioPerine® along with Curcuminoids. BioPerine® does not indiscriminatingly increase the Curcumin bioavailability. Other formulations increase the glucoronidation of Curcumin. In addition piperine has thermogenesis activity. As the thermogenesis occurs, the demand of fresh nutrients increases to sustain the metabolic process, facilitating better absorption of nutrients in intestine.
Safety of Curcumin C3 Complex® and BioPerine® has been evaluated in a clinical study performed by Tufts University. The results of the study showed that Curcumin and BioPerine® combination can be safely taken without any interaction with commonly prescribed medication such as flurbiprofen, acetaminophen and midazolam28. However no such data on drug interactions is available with any other curcumin formulation.
Extensive biotransformation in the body, an in-depth knowledge of Curcumin biotransformation and role of metabolites inside the body is important to apply the concept of bioavailability more constructively.
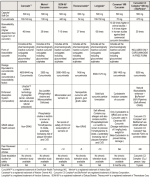
Table I gives an interesting comparison of above seven curcumin preparations in context of their composition, bioavailability, actual Curcumin in the product as well as estimated bioavailable Curcumin in the body.
The chart is informative for retailers & consumers to understand the difference between various curcumin preparations available in the market. The table can assist in choosing the right formulation by providing answers to following questions:
– Which formulation has maximum Curcumin in the product based on both composition as well as dose equivalency (Available Curcumin= Bioavailability Factor X content of Curcumin)
– What forms of Curcumin are made bioavailable (Does it include phase II metabolites?)
– Which formulation is GRAS and safe
– Use of excipients and associated toxicity
– How many clinical studies have been conducted on the formulations


































.gif)