i've been using AAS for years and never felt the need to take an AI since i was always into strength not looks and i never had gyno symptoms. i started taking MP arimidex @ 1mg eod, only 2 doses so far but i've noticed my redish color in my face and arms is gone(someone at work noticed it first) i can actually see seperation in some of my muscles now too. just thought i would share. can it be the arimidex doing this only after 2 doses??
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
arimidex?
- Thread starter JOEY11
- Start date
- Joined
- Jan 30, 2008
- Messages
- 556
Facial flushing in AAS users is a known side of improper estrodial, increased BP/hypertension, bloat, etc. AIs do help to lower and prevent these issues, quite significantly in some cases.
You'd be better off with Aromasin, to prevent the estro rebound, as well as the less harshness on your lipids, but at least you're working to use an AI now. The ole saying "live and learn" does apply here. Now you just need blood work to determine you're going in the right direction.
You'd be better off with Aromasin, to prevent the estro rebound, as well as the less harshness on your lipids, but at least you're working to use an AI now. The ole saying "live and learn" does apply here. Now you just need blood work to determine you're going in the right direction.
Last edited:
Facial flushing in AAS users is a known side of improper estrodial, increased BP/hypertension, bloat, etc. AIs do help to lower and prevent these issues, quite significantly in some cases.
You'd be better off with Aromasin, to prevent the estro rebound, as well as the less harshness on your lipids, but at least you're working to use an AI now. The ole saying "live and learn" does apply here. Now you just need blood work to determine you're going in the right direction.
thanks, i'll get some aromasin. 12.5mg ed should be good to start with
- Joined
- Feb 22, 2011
- Messages
- 790
That seems like a ton of arimidex 1mg EOD. I know people taking about 1g of test and other compliments getting good results on .25mg. MWF I guess I would just tell you to be careful you do not crash your esto level. Because then you will have a lot of joint pain and you libido will be completely gone. I would say start off lower and see how you feel and if you still feel like you are holding too much water then slowly dial it up. A much better approach. Remember Arimidex has a half life of about 3 days.
i wish i could but they are capsules, that's why i was taking EOD. i'll have some exemastane soon, they are dosed @ 12.5mg. should i take that EOD as well?
i'm currently on 700mg test, 100mg tren ed, 600 eq, will be dropping eq and adding winny @ 50mg ed. test bloats me bad, it makes me look like shit if i don't watch carbs and estro, just recently decided to change my bloated corpse look
i'm currently on 700mg test, 100mg tren ed, 600 eq, will be dropping eq and adding winny @ 50mg ed. test bloats me bad, it makes me look like shit if i don't watch carbs and estro, just recently decided to change my bloated corpse look
- Joined
- Jan 30, 2008
- Messages
- 556
AIs have been proven to increase your natural test 60-80%, yet another reason you should be using them while on AAS. 
I can only imagine how shut down your system is, however. You should also highly consider hCG, but please read the article in my sig in regards to taking NAC while using hCG.
I can only imagine how shut down your system is, however. You should also highly consider hCG, but please read the article in my sig in regards to taking NAC while using hCG.
- Joined
- Feb 22, 2011
- Messages
- 790
i wish i could but they are capsules, that's why i was taking EOD. i'll have some exemastane soon, they are dosed @ 12.5mg. should i take that EOD as well?
i'm currently on 700mg test, 100mg tren ed, 600 eq, will be dropping eq and adding winny @ 50mg ed. test bloats me bad, it makes me look like shit if i don't watch carbs and estro, just recently decided to change my bloated corpse look
You could always split the caps in half and take it that way. Also if you start the exemastane then you may be able to just get away with only taking it @ 12.5 ED I have heard a lot of people saying they would prefer the exem over arimidex. I have never tried it.
minister
Kilo Klub Member / Board Supporter
- Joined
- Sep 30, 2009
- Messages
- 1,621
1 mg of Adex EOD is way too much , unless you are trying to reverse gyno. like one of the guys say .25 EOD is sufficient, half life of Adex is long so that would be a good dose. Arimidex does not manage estrogen , it completely blocks it, you need estrogen for a lot of vital functions in the body from cholesterol balance to the muscle building process. God bless you. Minister.
- Joined
- Jan 30, 2008
- Messages
- 556
Actually, half-life of AIs in men is much less than in women. I was thinking 8-12 hours in men. I need to find that case study. Once I do I'll post it up.
- Joined
- Jan 30, 2008
- Messages
- 556
As I thought, here's the case study on Exemestane (Aromasin) in men:
**broken link removed**
Pharmacokinetics and Dose Finding of a Potent Aromatase Inhibitor, Aromasin (Exemestane), in Young Males
Abstract
Our friend heavyiron (heavyiron1 here, I believe) also discussed it in depth in a previous article he wrote (click the link below), which relied on data from the above case study, and others.
http://www.ironmagazineforums.com/anabolic-zone/106001-aromasin-exemestane.html
I feel it is more proper to dose AIs, at least Exemestane (Aromasin) ED, and not EOD. There would be too much fluctuation in EOD dosing. I would gander that Anastrozole (Arimidex) has a very similar halflife in men, as well.
**broken link removed**
Pharmacokinetics and Dose Finding of a Potent Aromatase Inhibitor, Aromasin (Exemestane), in Young Males
Abstract
Suppression of estrogen, via estrogen receptor or aromatase blockade, is being investigated in the treatment of different conditions. Exemestane (Aromasin) is a potent and selective irreversible aromatase inhibitor. To characterize its suppression of estrogen and its pharmacokinetic (PK) properties in males, healthy eugonadal subjects (14–26 yr of age) were recruited. In a cross-over study, 12 were randomly assigned to 25 and 50 mg exemestane daily, orally, for 10 d with a 14-d washout period. Blood was withdrawn before and 24 h after the last dose of each treatment period. A PK study was performed (n = 10) using a 25-mg dose. Exemestane suppressed plasma estradiol comparably with either dose [25 mg, 38% (P ≤ 0.002); 50 mg, 32% (P ≤ 0.008)], with a reciprocal increase in testosterone concentrations (60% and 56%; P ≤ 0.003 for both). Plasma lipids and IGF-I concentrations were unaffected by treatment. The PK properties of the 25-mg dose showed the highest exemestane concentrations 1 h after administration, indicating rapid absorption. The terminal half-life was 8.9 h. Maximal estradiol suppression of 62 ± 14% was observed at 12 h. The drug was well tolerated. In conclusion, exemestane is a potent aromatase inhibitor in men and an alternative to the choice of available inhibitors. Long-term efficacy and safety will need further study.
Our friend heavyiron (heavyiron1 here, I believe) also discussed it in depth in a previous article he wrote (click the link below), which relied on data from the above case study, and others.
http://www.ironmagazineforums.com/anabolic-zone/106001-aromasin-exemestane.html
I feel it is more proper to dose AIs, at least Exemestane (Aromasin) ED, and not EOD. There would be too much fluctuation in EOD dosing. I would gander that Anastrozole (Arimidex) has a very similar halflife in men, as well.
Last edited:
- Joined
- Jan 30, 2008
- Messages
- 556
Here's the post by heavyiron I linked to just above:
Aromasin
(Exemestane)
Aromasin is a steroidal aromatase inactivator used to lower circulating estrogen. It was developed to help fight breast cancer as estrogen plays a role in the growth of cancer cells. Aromasin binds irreversibly to the aromatase enzyme. This suppresses the conversion of androgens into estrogen. Circulating estrogen can be reduced by nearly 85% in women using Aromasin. A common misconception is that aromatase inhibition is similar in men than women. However in trials when males were administered 25mg of Aromasin daily maximal estradiol suppression of 62 ± 14% was observed at 12 hours. Aromasin acts as a false substrate for the aromatase enzyme, and is processed to an intermediate that binds irreversibly to the active site of the enzyme causing its inactivation, an effect also known as "suicide inhibition." In other words, Exemestane, by being structurally similar to the target of the enzymes, permanently binds to those enzymes, thereby preventing them from ever completing their task of converting androgens into estrogens. When we compare this mode of action against other AI’s the benefit becomes clear. Arimidex can unbind from the aromatase enzyme when you stop taking it but Aromasin will not therefore there is less chance of estrogen rebound with Aromasin.
Aromasin can be employed during a steroid cycle when aromatizing compounds such as testosterone are administered in order to control estrogen from getting out of control. During the course of a typical steroid cycle estrogen can rise quite high. Estrogen has been measured as much as 7 times higher than normal in men on steroids. This is excessive and can potentially cause water retention, gynecomastia (the formation of female breast tissue) or benign prostatic hyperplasia. Therefore in order to avoid these side effects estrogen must be controlled.
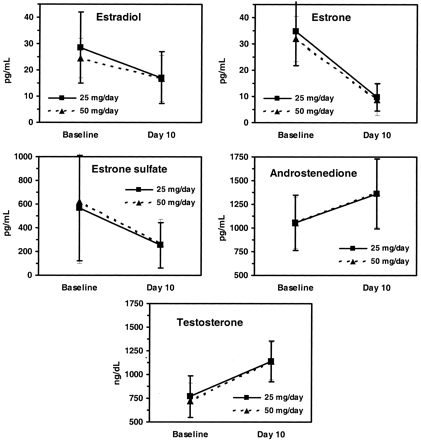
Aromasin not only lowers circulating estrogen and sex hormone binding globulin but it also increases free testosterone by a whopping 117%! Total testosterone increases about 60%. Check out the performance of Aromasin after just 10 days of treatment in males.

FIG. 1. Estrogen and androgen plasma levels after 10 d of daily exemestane (25 or 50 mg) in healthy young males (mean ± SD; n = 9–11). To convert to Systeme International units: estradiol, picomoles per liter (x3.671); estrone, picomoles per liter (x3.699); androstenedione, nanomoles per liter (*0.003492); and testosterone, nanomoles per liter (x0.03467).
Aromasin may be used during a steroid cycle with aromatizing compounds and during PCT to help keep the estrogen to testosterone balance in favor of testosterone. Out of all the medications to control estrogen, Aromasin seems to be the most well balanced. It raises testosterone slightly better than Arimidex and lowers estradiol about 12% better than arimidex in men and is likely to cause less estrogen rebound than Arimidex. Keep in mind that 50mg of Aromasin daily kept estradiol in the normal range for men so if you think using an aromatase inhibitor will crush estrogen too much this science supports the opposite. From the data I have read and my years of experience with this medication 25mg of Aromasin every other day is a good starting point on moderate doses of testosterone. If testosterone doses are raised then 25mg daily may be needed to control estrogen. Since either high and low estrogen can cause side effects such as low libido only labs can determine the appropriate dose of Aromasin.
Written by heavyiron
- Joined
- Jan 30, 2008
- Messages
- 556
I found a case study of using Arimidex (Anastrozole) in young males with onset of gynecomastia. I'll outline it below, and also compare the half-life graphs between it and the above Aromasin (Exemestane) case study in young males.
Again, here's the link to the Aromasin case study that I previously linked to above:
**broken link removed**
And here is the case study for Arimidex in boys:
**broken link removed**
Now, where one has to be careful with that above is statements of the terminal half-life of Arimidex in males being 46.8 hours. However, for a body-builder's particular use, when you look at the graphs (below) from both the case studies above of Arimidex and Aromasin half-lifes in males, you can clearly see a massive drop (approx. 50%) in circulating levels of the drug by 4 hours, and yet another big drop by the 8 hour mark. Granted, the Aromasin graph was based on 1 single treatment, versus the Arimidex measurement done after 14 days. However, the trends follow near exact on both, including the maximum absorption rate of approx. 1 hour for either drug.
It is important to note that Aromasin is a suicidal AI, whereas Arimidex is not. Therefore, Aromasin has potentially a longer effect to inhibit aromatase due to its irreversible mechanism.
**broken link removed**
Exemestane plasma concentrations vs. time in 10 young males receiving a single 25-mg oral dose.
**broken link removed**
Anastrozole plasma concentrations over time after 14 d oral administration of anastrozole 1 mg once daily (population = 36).
All in all, my personal feelings is that males taking an AI should do such ED (every day), and not EOD (every other day), which would keep the blood plasma levels more stable, and less chance of estrogen rebound effect. It is important that anyone using an AI to have blood work performed at least during and after cycle to ensure dosages are proper and ultimately safe.
Again, here's the link to the Aromasin case study that I previously linked to above:
**broken link removed**
And here is the case study for Arimidex in boys:
**broken link removed**
Pharmacokinetics and Pharmacodynamics of Anastrozole in Pubertal Boys with Recent-Onset Gynecomastia
Abstract
Context: Use of aromatase inhibitors to suppress estrogen production is being actively investigated in a variety of experimental conditions in both females and males. Anastrozole (Arimidex) is a potent and selective reversible inhibitor of the aromatase enzyme in females.
Objective: Our objective was to characterize the pharmacokinetics (PK) and pharmacodynamics (PD) of anastrozole in adolescent males with gynecomastia of less than 1 yr duration. The effect of anastrozole on breast size was also assessed as an exploratory aim.
Design: We conducted a PK/PD open-label study.
Setting: This clinical research center study was undertaken at pediatric academic centers.
Patients: Forty-two boys with gynecomastia (mean age 13 ± 1.8 yr; duration of gynecomastia 7.0 ± 2.5 months; body mass index 28.3 ± 5.9 kg/m2) were recruited.
Interventions: Anastrozole, 1 mg, was given daily for 6 months.
Main Outcomes: We assessed PK/PD of anastrozole after 14 d daily dosing and changes in breast size (exploratory aim) by manual tape measurements (area) and ultrasound (volume) after 6 months.
Results: Anastrozole was rapidly absorbed orally (time to reach maximum concentration, 1 h) with a slow apparent clearance of 1.54 liters/h and a terminal half-life of 46.8 h. Testosterone/estradiol ratios increased significantly with concomitant increase in LH/FSH concentrations indicating aromatase blockade. There was a reduction in breast area (∼63%) and breast volume (∼57%) in the study group as compared with baseline (P = 0.004). The drug was well tolerated.
Conclusions: Anastrozole is a potent aromatase inhibitor in adolescent males, with rapid absorption and slow elimination kinetics after oral dosing. Exploratory analysis of changes in breast size showed breast reduction in the cohort; this deserves further study.
Abstract
Context: Use of aromatase inhibitors to suppress estrogen production is being actively investigated in a variety of experimental conditions in both females and males. Anastrozole (Arimidex) is a potent and selective reversible inhibitor of the aromatase enzyme in females.
Objective: Our objective was to characterize the pharmacokinetics (PK) and pharmacodynamics (PD) of anastrozole in adolescent males with gynecomastia of less than 1 yr duration. The effect of anastrozole on breast size was also assessed as an exploratory aim.
Design: We conducted a PK/PD open-label study.
Setting: This clinical research center study was undertaken at pediatric academic centers.
Patients: Forty-two boys with gynecomastia (mean age 13 ± 1.8 yr; duration of gynecomastia 7.0 ± 2.5 months; body mass index 28.3 ± 5.9 kg/m2) were recruited.
Interventions: Anastrozole, 1 mg, was given daily for 6 months.
Main Outcomes: We assessed PK/PD of anastrozole after 14 d daily dosing and changes in breast size (exploratory aim) by manual tape measurements (area) and ultrasound (volume) after 6 months.
Results: Anastrozole was rapidly absorbed orally (time to reach maximum concentration, 1 h) with a slow apparent clearance of 1.54 liters/h and a terminal half-life of 46.8 h. Testosterone/estradiol ratios increased significantly with concomitant increase in LH/FSH concentrations indicating aromatase blockade. There was a reduction in breast area (∼63%) and breast volume (∼57%) in the study group as compared with baseline (P = 0.004). The drug was well tolerated.
Conclusions: Anastrozole is a potent aromatase inhibitor in adolescent males, with rapid absorption and slow elimination kinetics after oral dosing. Exploratory analysis of changes in breast size showed breast reduction in the cohort; this deserves further study.
Now, where one has to be careful with that above is statements of the terminal half-life of Arimidex in males being 46.8 hours. However, for a body-builder's particular use, when you look at the graphs (below) from both the case studies above of Arimidex and Aromasin half-lifes in males, you can clearly see a massive drop (approx. 50%) in circulating levels of the drug by 4 hours, and yet another big drop by the 8 hour mark. Granted, the Aromasin graph was based on 1 single treatment, versus the Arimidex measurement done after 14 days. However, the trends follow near exact on both, including the maximum absorption rate of approx. 1 hour for either drug.
It is important to note that Aromasin is a suicidal AI, whereas Arimidex is not. Therefore, Aromasin has potentially a longer effect to inhibit aromatase due to its irreversible mechanism.
**broken link removed**
Exemestane plasma concentrations vs. time in 10 young males receiving a single 25-mg oral dose.
**broken link removed**
Anastrozole plasma concentrations over time after 14 d oral administration of anastrozole 1 mg once daily (population = 36).
All in all, my personal feelings is that males taking an AI should do such ED (every day), and not EOD (every other day), which would keep the blood plasma levels more stable, and less chance of estrogen rebound effect. It is important that anyone using an AI to have blood work performed at least during and after cycle to ensure dosages are proper and ultimately safe.
Last edited:
Similar threads
- Replies
- 2
- Views
- 339
Popular tags
aas
aas testing
anabolic steroids
anabolics online
anabolid steroids
anadrol
anadrol drol tabs inj
anavar
anavar and winnie
body building
body building supplements
bodybuilder
bodybuilding
bodybuilding steroid test
clenbuterol
cycle
deca tren dosage
deca-durobolin
dianabol
dianabol and oxy
gear
hcg
hgh
motivation
muscle building
muscle mass
nandrolone
oxandrolone
pct
peptides
quality raw
raw steroid powders
steroid cycle
steroids
suspension
sust300
sustanon
test
test 400
test ace
test cyp
test cypionate
test e
test prop
testosterone
testosterone boosters
testosterone cypionate
testsuspension
tren ace
trenbolone acetate
Popular tags
aas
aas testing
anabolic steroids
anabolics online
anabolid steroids
anadrol
anadrol drol tabs inj
anavar
anavar and winnie
body building
body building supplements
bodybuilder
bodybuilding
bodybuilding steroid test
clenbuterol
cycle
deca tren dosage
deca-durobolin
dianabol
dianabol and oxy
gear
hcg
hgh
motivation
muscle building
muscle mass
nandrolone
oxandrolone
pct
peptides
quality raw
raw steroid powders
steroid cycle
steroids
suspension
sust300
sustanon
test
test 400
test ace
test cyp
test cypionate
test e
test prop
testosterone
testosterone boosters
testosterone cypionate
testsuspension
tren ace
trenbolone acetate
Members online
- Fullblown
- dxteran
- P30LEM
- demon
- Onidus
- Jammin
- Seattlesbest
- Dlrob27
- iwantmass
- trapper john
- slesh
- bizzbtw
- Multiplex
- Bleed
- airman
- hk55874
- liv2pb
- skinnyrunt
- TrippplePPP
- Bulldog1
- hevi-head
- Reno911
- LFG
- Nicky_Slim24
- nothuman
- Nlargedshadow
- Cerberus777
- MackCoach
- GiantMuscle
- luki7788
- Heath82
- Bodyofwork365
- bigstick
- mhpump
- mike036
- John Jeeves
- duckdodger2
- powerforward
- iceman92
- juiceddunk2.0
- K Bomb
- Drax
- pistonpump
- he-man100
- DIRECT
- btails
- maxrep24
- scooped
- SOUR DIESEL
- hutechlab
Total: 1,322 (members: 1,312, guests: 10)
Forum statistics
- Total page views
- 559,655,996
- Threads
- 136,131
- Messages
- 2,780,510
- Members
- 160,446
- Latest member
- ctrcivic


































.gif)