- Joined
- Jul 25, 2008
- Messages
- 1,700
Hey dat I read that study you posted (pg 10 AM) called:
"GHRP-2, a GHS-R agonist, directly acts on myocytes to attenuate the dexamethasone-induced expressions of muscle-specific ubiquitin ligases, Atrogin-1 and MuRF1, Daisuke Yamamoto, et al., Life Sciences 82 (2008) 460–466"
vast568; said:Is there any way you could break this down into us simple people language? I really do not understand " Atrogin-1 and MuRF1 mRNA" but it seems like GHRP-2 and 6 may be more different than one just not causing hunger. Thanks.
The primary research in this area is focused on preventing muscle wasting due to age, inactivity, disease & medical states such as AIDS, cancer, sepsis & burn victims...
...so they explore the molecular mechanisms modulating muscle mass.
There are two ways to do that. One increase HYPERTROPHY, the other decrease ATROPHY.
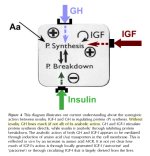
These are not two sides of the same coin. A primary mode for hypertrophy is understanding the IGF-1 signaling. This is what takes place once an IGF-1 molecule binds to a IGF Receptor in target tissue. A decent mental image is a lock (receptor) accepts the key (IGF-1 molecule) which turns the switch and starts a cascade of signaling which results in blocking apoptosis (cell death), inducing protein synthesis, gene transcription and proliferation. All of this induces skeletal muscle hypertrophy.
All of us experience atrophy to some dgree but it is most pronounced if we are vastly underfed, have a severe hormonal imbalance or have an underlying medical condition. In those instances a process of muscle atrophy accelerates. Conditions that cause atrophy lead to a decrease in the size of pre-existing muscle fibers resulting from increases in the rate of ATP-dependent ubiquitin-mediated proteolysis.
Proteolysis is the degradation (digestion) of proteins. Ubiquitin is a regulatory protein whose primary function is labeling proteins for degradation. The labeling process is catalyzed by enzymes called ubiquitin ligases. Once this process is initiated other ligases attach additional ubiquitin molecules to create a polyubiquitin chain. This chain is easily bound by and eventually degraded by a proteasome.
That is how muscle fiber is digested.
Where do these highly destructive ubiquitin-protein ligases come from?
There are genes that encode these proteins. Normally a certain amount is created but under conditions of atrophy these genes may be very active and encode/create a lot of these destructive ligases.
Those genes were identified within the last 5 or so years and they are:
MAFbx/Atrogin-1, and MuRF1
In mice it was discovered that these two genes are required for muscle atrophy because muscle atrophy doesn't occur in mice if these genes are knocked out.
So some research focuses on ways to reduce these two genes expression during periods when naturally they would become most active.
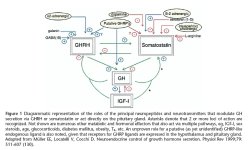
Enter our friends the "Growth Hormone Releasing Peptides" and the studies you mention posted in my thread on AnabolicMinds.
Last edited:


































.gif)