- Joined
- Apr 9, 2021
- Messages
- 1,724
Clenbuterol focus: Research on Clen & Beta2- Adrenergic Compounds, Protocols
Author: Type-IIx
Research considerations
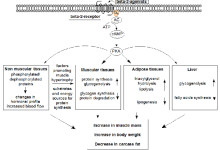
Clenbuterol is a drug legally used in human medicine in a select few countries as a bronchodilator at doses up to 40µg daily [18]. Primarily, it is used illicitly as a potent repartitioning agent to promote growth in cattle and sheep by increasing protein accretion and fat removal with little or no change in body weight, and by human athletes for this effect [8].
This work thus makes some extrapolation from animal research on clenbuterol as well as from human research on similar compounds (i.e., salbutamol/albuterol, terbutaline).
Pharmacokinetics
Clenbuterol is a β₂-adrenergic agonist similar in some structural respects to salbutamol (albuterol). Agonism of the β₂ receptor stimulates adenylyl cyclase activity which ultimately leads to downstream effects of smooth muscle relaxation in the bronchioles as a therapeutic target. Clenbuterol is a potent sympathomimetic.
Oral bioavailability of 70-80% and a long half-life of 25-39 hours [6].
Desensitization
Tachyphylaxis, or desensitization, is a feature of the β₂AR. This is likely because β₂AR activation and stimulation of downstream effects are a target for phosphorylation (50) and/or because it binds β-arrestin (51), an accessory protein involved in G protein-coupled receptor desensitization (52) [16].
Tolerability
High doses of clenbuterol have been used in the literature. The highest doses used are for patients with LVAD (left ventricular atrioventricular devices) and congestive heart failure. For this purpose, clenbuterol is used specifically for its effects on cardiac remodeling! It is important to note that cardiac remodeling should generally be avoided by healthy persons (though the remodeling from clen is ostensibly not maladaptive)! There was no change in LV mass with 80 µg daily for 3 months, [19].
Otherwise, an important research consideration is that tolerability cannot be assessed nor inferred from dosages administered in studies. Rather, tolerability must be assessed as an endpoint in the trial, in order to be able to make a claim regarding the drug's tolerability.
Here, this author presents some actual data on tolerability where it has been assessed as an end-point.
From a study on patients with heart failure. Here, the dosage started at 20µg twice daily and was titrated up to 40µg twice daily after a week [19]. Two of the nineteen (2/19) subjects dropped out of the 3 months-long study due to clenbuterol side effects:
Two clenbuterol subjects required discontinuation of study drug (asymptomatic slow ventricular tachycardia, severe muscle cramps without significant elevation increatine kinase [CK]) [19].
One further clenbuterol subject had a high rate of ventricular ectopy that disappeared without reduction in the clenbuterol dose [19].
One placebo subject had frequent non-sustained ventricular tachycardia. There were no implantable cardioverter-defibrillator discharges during the course of the study [19].
Six clenbuterol and 2 placebo subjects reported mild muscle cramps [19].
The CK value was elevated in 5 clenbuterol and 4 placebo subjects. The range of peak CK was 300 to 597 mg/dl in clenbuterol subjects and 305 to 408 mg/dl with placebo. Three clenbuterol subjects had cramps without elevation of CK, and CK was elevated in 1 clenbuterol subject who was asymptomatic. Of importance was that the CK level decreased despite continued drug administration (Figure 1B) [19].
Tremors were reported in 5 clenbuterol and 2 placebo subjects [19].
In a large, double-blind clinical trial (n=175 women with stress incontinence, 82 received 40µg clen daily for 2 weeks):
Side effects were noted in 13 clenbuterol-administered patients (15.9%), and the treatment was discontinued in 5 of these (due to tremors of the finger in 2 cases; dizziness in 1; urinary hesitancy in 1; and loss of appetite in 1). In the remaining 8 patients the side effects were mild and the treatment was able to be completed. In the clenbuterol group, the main side effect was finger tremors (8 out of 82, 9.8%), and tachycardia was also noted (2 out of 82, 2.4%) [20].
In the placebo group, side effects were noted in 12 patients (12.9%), and the treatment was discontinued in 4 of these (dizziness in two patients and nausea in two. One of these 4 patients was treated for longer than 1 week, and the data were thus included in the statistical analysis). In the placebo group, the main side effects were gastrointestinal disturbance (7 out of 93, 7.5%) and dizziness (3 out of 93, 3.2%) [20].
Side effects
- Electrolyte disturbance (primarily hypokalemia and hyperglycemia) and associated muscle cramping
- Tachycardia
- Dyskinesia
- Tremor
- Liver failure
- Muscle atrophy
- Myocardial infarction
- Myocardial reperfusion injury
Cardiac effects
The infamous rat study
"myotoxic" cardiac necrosis
Provides evidence of cardiotoxicity with high doses in rats:
Burrinston et al. (21) explored the myotoxic effects of clenbuterol injected subcutaneously. Rats were treated with a single shot of clenbuterol. The amount of necrosis in the muscle was measured using an anti-myosin monoclonal antibody that enters the endoplasmic reticulum only in cells that have undergone necrosis. The amount of necrosis in the heart was positively correlated with the amount of clenbuterol that was injected. Necrosis was seen at 4 hours post administration and peaked at 15 hours. This finding can explain the cardiomyocyte damage seen after a single ingestion of clenbuterol. Necrosis decreased by 91-100% in rats pre-treated with bisoprolol, β2 blockade or noradrenaline depletion. This finding is consistent with direct myotoxic effect of clenbuterol. Peak cardiomyocyte necrosis was noted 2.4 mm from the apex. This finding is consistent with the high incidence of ECG and echocardiographic changes in the in the inferior and lateral wall in the patients presented. Direct myocardial injury due to catecholamine activation has been well described and attributed to supply/demand imbalance induced by the sustained activation of adrenergic receptors and mitochondrial dysfunction (22) [6].
These pathological changes are not to be taken lightly. Clen causes real cardiac harm at performance- and physique- enhancement dosages.
Interestingly,
Case reports of real myocardial injury
See [6].
A common theme of absurdly high doses.
Mechanisms of myocardial injury
cardiac necrosis
[Clenbuterol-induced myocyte necrosis] appear to be driven through clenbuterol-induced pre-synaptic release of catecholamines within the myocardium which cause myocyte damage through a β1-specific mechanism. Such effects can be blocked in vivo by the use of β1-specific antagonists [e.g., bisoprolol] [9].
apoptosis = cell death
The lowest dose of clenbuterol to induce cardiomyocyte apoptosis was 1 microg/kg, with peak apoptosis (0.35 +/- 0.05%; P < 0.05) occurring in response to 5 mg/kg. In the soleus, peak apoptosis (5.8 +/- 2%; P < 0.05) was induced by the lower dose of 10 microg/kg. Cardiomyocyte apoptosis was detected throughout the ventricles, atria, and papillary muscles. However, this damage was most abundant in the left ventricular subendocardium at a point 1.6 mm, that is, approximately one-quarter of the way, from the apex toward the base. beta-AR antagonism (involving propranolol, bisoprolol, or ICI 118551) or reserpine was used to show that clenbuterol-induced myocardial apoptosis was mediated through neuromodulation of the sympathetic system and the cardiomyocyte beta1-AR...
[15]
cardiac hypertrophy
In rat cardiocytes (heart cells), Clenbuterol is associated with a threefold increase in IGF-I mRNA expression. An increase in ANP, BNP, but not αSkA indicates physiological cardiac hypertrophy. Cardiac fibroblasts contain essentially only β2 receptors and are the prime targets of clenbuterol indicating a paracrine signalling role for physiological cardiac hypertrophy. Hypothesis that the β₂-AR physiological function is myocardial protection against stress [9].
Author: Type-IIx
Research considerations
Clenbuterol is a drug legally used in human medicine in a select few countries as a bronchodilator at doses up to 40µg daily [18]. Primarily, it is used illicitly as a potent repartitioning agent to promote growth in cattle and sheep by increasing protein accretion and fat removal with little or no change in body weight, and by human athletes for this effect [8].
This work thus makes some extrapolation from animal research on clenbuterol as well as from human research on similar compounds (i.e., salbutamol/albuterol, terbutaline).
Pharmacokinetics
Clenbuterol is a β₂-adrenergic agonist similar in some structural respects to salbutamol (albuterol). Agonism of the β₂ receptor stimulates adenylyl cyclase activity which ultimately leads to downstream effects of smooth muscle relaxation in the bronchioles as a therapeutic target. Clenbuterol is a potent sympathomimetic.
Oral bioavailability of 70-80% and a long half-life of 25-39 hours [6].
Desensitization
Tachyphylaxis, or desensitization, is a feature of the β₂AR. This is likely because β₂AR activation and stimulation of downstream effects are a target for phosphorylation (50) and/or because it binds β-arrestin (51), an accessory protein involved in G protein-coupled receptor desensitization (52) [16].
Tolerability
High doses of clenbuterol have been used in the literature. The highest doses used are for patients with LVAD (left ventricular atrioventricular devices) and congestive heart failure. For this purpose, clenbuterol is used specifically for its effects on cardiac remodeling! It is important to note that cardiac remodeling should generally be avoided by healthy persons (though the remodeling from clen is ostensibly not maladaptive)! There was no change in LV mass with 80 µg daily for 3 months, [19].
Otherwise, an important research consideration is that tolerability cannot be assessed nor inferred from dosages administered in studies. Rather, tolerability must be assessed as an endpoint in the trial, in order to be able to make a claim regarding the drug's tolerability.
Here, this author presents some actual data on tolerability where it has been assessed as an end-point.
From a study on patients with heart failure. Here, the dosage started at 20µg twice daily and was titrated up to 40µg twice daily after a week [19]. Two of the nineteen (2/19) subjects dropped out of the 3 months-long study due to clenbuterol side effects:
Clenbuterol at 80µg/day was well tolerated [19].
Two clenbuterol subjects required discontinuation of study drug (asymptomatic slow ventricular tachycardia, severe muscle cramps without significant elevation increatine kinase [CK]) [19].
One further clenbuterol subject had a high rate of ventricular ectopy that disappeared without reduction in the clenbuterol dose [19].
One placebo subject had frequent non-sustained ventricular tachycardia. There were no implantable cardioverter-defibrillator discharges during the course of the study [19].
Six clenbuterol and 2 placebo subjects reported mild muscle cramps [19].
The CK value was elevated in 5 clenbuterol and 4 placebo subjects. The range of peak CK was 300 to 597 mg/dl in clenbuterol subjects and 305 to 408 mg/dl with placebo. Three clenbuterol subjects had cramps without elevation of CK, and CK was elevated in 1 clenbuterol subject who was asymptomatic. Of importance was that the CK level decreased despite continued drug administration (Figure 1B) [19].
Tremors were reported in 5 clenbuterol and 2 placebo subjects [19].
In a large, double-blind clinical trial (n=175 women with stress incontinence, 82 received 40µg clen daily for 2 weeks):
Side effects were noted in 13 clenbuterol-administered patients (15.9%), and the treatment was discontinued in 5 of these (due to tremors of the finger in 2 cases; dizziness in 1; urinary hesitancy in 1; and loss of appetite in 1). In the remaining 8 patients the side effects were mild and the treatment was able to be completed. In the clenbuterol group, the main side effect was finger tremors (8 out of 82, 9.8%), and tachycardia was also noted (2 out of 82, 2.4%) [20].
In the placebo group, side effects were noted in 12 patients (12.9%), and the treatment was discontinued in 4 of these (dizziness in two patients and nausea in two. One of these 4 patients was treated for longer than 1 week, and the data were thus included in the statistical analysis). In the placebo group, the main side effects were gastrointestinal disturbance (7 out of 93, 7.5%) and dizziness (3 out of 93, 3.2%) [20].
Side effects
- Electrolyte disturbance (primarily hypokalemia and hyperglycemia) and associated muscle cramping
- Tachycardia
- Dyskinesia
- Tremor
- Liver failure
- Muscle atrophy
- Myocardial infarction
- Myocardial reperfusion injury
Cardiac effects
The infamous rat study
"myotoxic" cardiac necrosis
Provides evidence of cardiotoxicity with high doses in rats:
Burrinston et al. (21) explored the myotoxic effects of clenbuterol injected subcutaneously. Rats were treated with a single shot of clenbuterol. The amount of necrosis in the muscle was measured using an anti-myosin monoclonal antibody that enters the endoplasmic reticulum only in cells that have undergone necrosis. The amount of necrosis in the heart was positively correlated with the amount of clenbuterol that was injected. Necrosis was seen at 4 hours post administration and peaked at 15 hours. This finding can explain the cardiomyocyte damage seen after a single ingestion of clenbuterol. Necrosis decreased by 91-100% in rats pre-treated with bisoprolol, β2 blockade or noradrenaline depletion. This finding is consistent with direct myotoxic effect of clenbuterol. Peak cardiomyocyte necrosis was noted 2.4 mm from the apex. This finding is consistent with the high incidence of ECG and echocardiographic changes in the in the inferior and lateral wall in the patients presented. Direct myocardial injury due to catecholamine activation has been well described and attributed to supply/demand imbalance induced by the sustained activation of adrenergic receptors and mitochondrial dysfunction (22) [6].
These pathological changes are not to be taken lightly. Clen causes real cardiac harm at performance- and physique- enhancement dosages.
Interestingly,
[9].[Clenbuterol-induced myocyte necrosis] appear to be driven through clenbuterol-induced pre-synaptic release of catecholamines within the myocardium which cause myocyte damage through a β1-specific mechanism. Such effects can be blocked in vivo by the use of β1-specific antagonists [e.g., bisoprolol].
Case reports of real myocardial injury
See [6].
A common theme of absurdly high doses.
Mechanisms of myocardial injury
cardiac necrosis
[Clenbuterol-induced myocyte necrosis] appear to be driven through clenbuterol-induced pre-synaptic release of catecholamines within the myocardium which cause myocyte damage through a β1-specific mechanism. Such effects can be blocked in vivo by the use of β1-specific antagonists [e.g., bisoprolol] [9].
apoptosis = cell death
The lowest dose of clenbuterol to induce cardiomyocyte apoptosis was 1 microg/kg, with peak apoptosis (0.35 +/- 0.05%; P < 0.05) occurring in response to 5 mg/kg. In the soleus, peak apoptosis (5.8 +/- 2%; P < 0.05) was induced by the lower dose of 10 microg/kg. Cardiomyocyte apoptosis was detected throughout the ventricles, atria, and papillary muscles. However, this damage was most abundant in the left ventricular subendocardium at a point 1.6 mm, that is, approximately one-quarter of the way, from the apex toward the base. beta-AR antagonism (involving propranolol, bisoprolol, or ICI 118551) or reserpine was used to show that clenbuterol-induced myocardial apoptosis was mediated through neuromodulation of the sympathetic system and the cardiomyocyte beta1-AR...
[15]
cardiac hypertrophy
In rat cardiocytes (heart cells), Clenbuterol is associated with a threefold increase in IGF-I mRNA expression. An increase in ANP, BNP, but not αSkA indicates physiological cardiac hypertrophy. Cardiac fibroblasts contain essentially only β2 receptors and are the prime targets of clenbuterol indicating a paracrine signalling role for physiological cardiac hypertrophy. Hypothesis that the β₂-AR physiological function is myocardial protection against stress [9].



































.gif)