Newer research. Full text in the link.
Background and aimsHyperglycemia and insulin resistance are concerns today worldwide. Recently, L-carnitine supplementation has been suggested as an effective adjunctive therapy in glycemic control. Therefore, it seems important to investigate its effect on glycemic markers.MethodsPubMed...

www.frontiersin.org
The effects of L-carnitine supplementation on glycemic markers in adults: A systematic review and dose-response meta-analysis
Background and aims: Hyperglycemia and insulin resistance are concerns today worldwide. Recently, L-carnitine supplementation has been suggested as an effective adjunctive therapy in glycemic control. Therefore, it seems important to investigate its effect on glycemic markers.
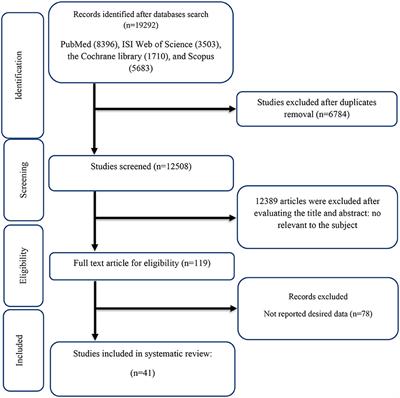
Methods: PubMed, Scopus, Web of Science, and the Cochrane databases were searched in October 2022 for prospective studies on the effects of L-carnitine supplementation on glycemic markers. Inclusion criteria included adult participants and taking oral L-carnitine supplements for at least seven days. The pooled weighted mean difference (WMD) was calculated using a random-effects model.
Results: We included the 41 randomized controlled trials (RCTs) (
n = 2900) with 44 effect sizes in this study. In the pooled analysis; L-carnitine supplementation had a significant effect on fasting blood glucose (FBG) (mg/dl) [WMD = −3.22 mg/dl; 95% CI, −5.21 to −1.23;
p = 0.002;
I2 = 88.6%,
p < 0.001], hemoglobin A1c (HbA1c) (%) [WMD = −0.27%; 95% CI, −0.47 to −0.07;
p = 0.007;
I2 = 90.1%,
p < 0.001] and homeostasis model assessment-estimate insulin resistance (HOMA-IR) [WMD = −0.73; 95% CI, −1.21 to −0.25;
p = 0.003;
I2 = 98.2%,
p < 0.001] in the intervention compared to the control group. L-carnitine supplementation had a reducing effect on baseline FBG ≥100 mg/dl, trial duration ≥12 weeks, intervention dose ≥2 g/day, participants with overweight and obesity (baseline BMI 25–29.9 and >30 kg/m2), and diabetic patients. Also, L-carnitine significantly affected insulin (pmol/l), HOMA-IR (%), and HbA1c (%) in trial duration ≥12 weeks, intervention dose ≥2 g/day, and participants with obesity (baseline BMI >30 kg/m2). It also had a reducing effect on HOMA-IR in diabetic patients, non-diabetic patients, and just diabetic patients for insulin, and HbA1c. There was a significant nonlinear relationship between the duration of intervention and changes in FBG, HbA1c, and HOMA-IR. In addition, there was a significant nonlinear relationship between dose (≥2 g/day) and changes in insulin, as well as a significant linear relationship between the duration (weeks) (coefficients = −16.45,
p = 0.004) of intervention and changes in HbA1C.


































.gif)