- Joined
- Apr 9, 2021
- Messages
- 1,718
I've noticed a paucity of decent articles on T3/T4. I've also seen some mention of side effects like palpitations/heart rate that was not attributed to T3 use, when it probably should have been. So, I think a thread on T3 and T4, some ramifications, including rhGH, is a good idea.
Definitions:
T3: Triiodothyronine
T3 (exogenous) [Cytomel, LT3]: Liothyronine
T4: Thyroxine
T4 (exogenous) [Tiromel, LT4]: Levothyroxine
T4's effects on resting energy expenditure/body composition:
[Effects of Altering Levothyroxine Dose on Energy Expenditure and Body Composition in Subjects Treated With LT4]
Tying this together with the pertinent Cytomel (Liothyronine) findings:

This is the average time-course for fT3 in healthy euthyroid adults given a single 50µg oral dose of Cytomel (Liothyronine) [1].
This is just an average, one PK paper from Goede et al. shows how individualized responses are, see [2].
mean half-life (t1/2) ≈ 1 d (22 h [1])
Given a 50 µg Cytomel dose:
Dose-response
Given a 75 µg Cytomel dose (daily for 14 days):
- fT3 increased by 1.7-fold
- RMR increased by 15% (adjusted for LBM)
- UCP2 increased 1.7-fold and UCP3 increased 2.4-fold [4]
Mechanism of action
The primary mechanism by which T4 (exogenous, Tiromel) increases RMR in humans is through conversion to T3.
The primary mechanism by which T3 increases RMR in humans is not through transcriptional control of the genes encoding mitochondrial proteins [4]. In rats, it is clear that T3 works by uncoupling electron transport from ATP synthesis via increased mRNA expression and mitochondrial protein levels of UCP-3 [3]. UCP-3 is principally expressed in skeletal muscle and heart tissue (in addition to brown and white adipose tissue) [3].
... after T3 injection the variations with time in UCP3 mitochondrial protein content coincided closely with the induced changes in RMR [3].
In rats, evidence of mitochondrial uncoupling was an increase in the nonphosphorylating respiration rate accompanied by a decrease in membrane potential (ΔΨ) of 40% and 8%, respectively [3].
Conversely, in humans, T3 increases RMR/REE without altering the respiratory chain transcription program, as evidenced by a lack of COX2 and mtTFA mRNA induction [4]. It definitely increases mRNA expression of UCP3 and UCP2, and resulted in a significant (15% x 75 µg) RMR/REE increase [4].
Risks, complications
Liothyronine (Cytomel) & Levothyroxine (Tiromel) are associated with increased cardiac arrhythmia events; and may lead to (drug-induced) thyrotoxicosis.
Overt thyrotoxicosis is defined as elevated serum free thyroxine (FT4) and free triiodothyronine (FT3), and suppressed thyrotropin (TSH) concentrations. Thyrotoxicosis with TSH suppression only (TTSO), and normal thyroid hormone concentrations, is also defined as mild thyrotoxicosis. Both overt thyrotoxicosis and TTSO may be caused by the same thyroid disorders. The most common cause of thyrotoxicosis is the use of excessive doses of L-thyroxine for the treatment of hypothyroidism.
Classic symptoms of thyrotoxicosis include:
rhGH and T4
Though therapeutic use of rhGH does not cause central hypothyroidism, it may reveal it. A primary mechanism in rhGH's lipolysis is the peripheral conversion of T4 to T3. There are, also, no long-term studies of rhGH in supra-physiological doses. According to at least one well-respected bodybuilding coach ( @luki7788 ), symptoms of hypothyroidism do indeed present with chronic, supra-physiological administration.
The symptoms of hypothyroidism to watch for would be:
There are other symptoms of hypothyroidism; however, they are easily confused with primary effects of rhGH and/or AAS use.
_______________________________________
References:
[1] Jonklaas, J., Burman, K. D., Wang, H., & Latham, K. R. (2015). Single-Dose T3 Administration. Therapeutic Drug Monitoring, 37(1), 110–118. doi:10.1097/ftd.0000000000000113
[2] GOEDE, S. L., LATHAM, K. R., LEOW, M. K.-S., & JONKLAAS, J. (2017). HIGH RESOLUTION FREE TRIIODOTHYRONINE-THYROTROPIN (FT3-TSH) RESPONSES TO A SINGLE ORAL DOSE OF LIOTHYRONINE IN HUMANS: EVIDENCE OF DISTINCT INTER-INDIVIDUAL DIFFERENCES UNRAVELED USING AN ELECTRICAL NETWORK MODEL. Journal of Biological Systems, 25(01), 119–143. doi:10.1142/s0218339017500073
[3] De Lange, P., Lanni, A., Beneduce, L., Moreno, M., Lombardi, A., Silvestri, E., & Goglia, F. (2001). Uncoupling Protein-3 Is a Molecular Determinant for the Regulation of Resting Metabolic Rate by Thyroid Hormone. Endocrinology, 142(8), 3414–3420. doi:10.1210/endo.142.8.8303
[4] BARBE, P., LARROUY, D., BOULANGER, C., CHEVILLOTTE, E., VIGUERIE, N., THALAMAS, C., … LANGIN, D. (2001). Triiodothyronine-mediated up-regulation of UCP2 and UCP3 mRNA expression in human skeletal muscle without coordinated induction of mitochondrial respiratory chain genes . The FASEB Journal, 15(1), 13–15. doi:10.1096/fj.00-0502fje
[5] Baumgartner, C., da Costa, B. R., Collet, T.-H., Feller, M., Floriani, C., Bauer, D. C., … Rodondi, N. (2017). Thyroid Function Within the Normal Range, Subclinical Hypothyroidism, and the Risk of Atrial Fibrillation. Circulation, 136(22), 2100–2116. doi:10.1161/circulationaha.117.028753
Definitions:
T3: Triiodothyronine
T3 (exogenous) [Cytomel, LT3]: Liothyronine
T4: Thyroxine
T4 (exogenous) [Tiromel, LT4]: Levothyroxine
T4's effects on resting energy expenditure/body composition:
[Effects of Altering Levothyroxine Dose on Energy Expenditure and Body Composition in Subjects Treated With LT4]
A trend was also seen for a direct correlation between REE/LBM and increases in fT3 levels, with an average 0.11 kcal/kg/d increase in REE/LBM for each 10 pg/dL increase in fT3.
Tying this together with the pertinent Cytomel (Liothyronine) findings:
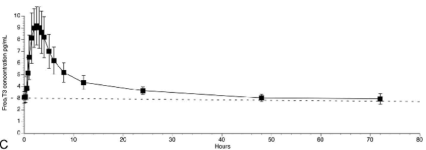
This is the average time-course for fT3 in healthy euthyroid adults given a single 50µg oral dose of Cytomel (Liothyronine) [1].
This is just an average, one PK paper from Goede et al. shows how individualized responses are, see [2].
mean half-life (t1/2) ≈ 1 d (22 h [1])
Given a 50 µg Cytomel dose:
- TSH change inverse to fT3, nadir 12 h, remained below baseline for ~ 72 h
- TSH begins to escape from suppression at a T3 concentration ≈ 153 ng/dL with a gradual recovery over several days; about 96 h before TSH returns to baseline with a 100µg dose (18)
- temp. increased insignificantly (p = 0.56)
- mean HR ↑ 18 bpm within 12 h [1]
Dose-response
Given a 75 µg Cytomel dose (daily for 14 days):
- fT3 increased by 1.7-fold
- RMR increased by 15% (adjusted for LBM)
- UCP2 increased 1.7-fold and UCP3 increased 2.4-fold [4]
Mechanism of action
The primary mechanism by which T4 (exogenous, Tiromel) increases RMR in humans is through conversion to T3.
The primary mechanism by which T3 increases RMR in humans is not through transcriptional control of the genes encoding mitochondrial proteins [4]. In rats, it is clear that T3 works by uncoupling electron transport from ATP synthesis via increased mRNA expression and mitochondrial protein levels of UCP-3 [3]. UCP-3 is principally expressed in skeletal muscle and heart tissue (in addition to brown and white adipose tissue) [3].
... after T3 injection the variations with time in UCP3 mitochondrial protein content coincided closely with the induced changes in RMR [3].
In rats, evidence of mitochondrial uncoupling was an increase in the nonphosphorylating respiration rate accompanied by a decrease in membrane potential (ΔΨ) of 40% and 8%, respectively [3].
Conversely, in humans, T3 increases RMR/REE without altering the respiratory chain transcription program, as evidenced by a lack of COX2 and mtTFA mRNA induction [4]. It definitely increases mRNA expression of UCP3 and UCP2, and resulted in a significant (15% x 75 µg) RMR/REE increase [4].
Risks, complications
Liothyronine (Cytomel) & Levothyroxine (Tiromel) are associated with increased cardiac arrhythmia events; and may lead to (drug-induced) thyrotoxicosis.
Overt thyrotoxicosis is defined as elevated serum free thyroxine (FT4) and free triiodothyronine (FT3), and suppressed thyrotropin (TSH) concentrations. Thyrotoxicosis with TSH suppression only (TTSO), and normal thyroid hormone concentrations, is also defined as mild thyrotoxicosis. Both overt thyrotoxicosis and TTSO may be caused by the same thyroid disorders. The most common cause of thyrotoxicosis is the use of excessive doses of L-thyroxine for the treatment of hypothyroidism.
Classic symptoms of thyrotoxicosis include:
- heat intolerance
- palpitations
- anxiety
- fatigue
- weight loss, and
- tremor
rhGH and T4
Though therapeutic use of rhGH does not cause central hypothyroidism, it may reveal it. A primary mechanism in rhGH's lipolysis is the peripheral conversion of T4 to T3. There are, also, no long-term studies of rhGH in supra-physiological doses. According to at least one well-respected bodybuilding coach ( @luki7788 ), symptoms of hypothyroidism do indeed present with chronic, supra-physiological administration.
The symptoms of hypothyroidism to watch for would be:
- dry and scaly skin
- sensitivity to cold
- brittle hair and nails
- slow movements and thoughts
- depression
- Only if not using clen, muscle cramps may be considered as an additional symptom
There are other symptoms of hypothyroidism; however, they are easily confused with primary effects of rhGH and/or AAS use.
_______________________________________
References:
[1] Jonklaas, J., Burman, K. D., Wang, H., & Latham, K. R. (2015). Single-Dose T3 Administration. Therapeutic Drug Monitoring, 37(1), 110–118. doi:10.1097/ftd.0000000000000113
[2] GOEDE, S. L., LATHAM, K. R., LEOW, M. K.-S., & JONKLAAS, J. (2017). HIGH RESOLUTION FREE TRIIODOTHYRONINE-THYROTROPIN (FT3-TSH) RESPONSES TO A SINGLE ORAL DOSE OF LIOTHYRONINE IN HUMANS: EVIDENCE OF DISTINCT INTER-INDIVIDUAL DIFFERENCES UNRAVELED USING AN ELECTRICAL NETWORK MODEL. Journal of Biological Systems, 25(01), 119–143. doi:10.1142/s0218339017500073
[3] De Lange, P., Lanni, A., Beneduce, L., Moreno, M., Lombardi, A., Silvestri, E., & Goglia, F. (2001). Uncoupling Protein-3 Is a Molecular Determinant for the Regulation of Resting Metabolic Rate by Thyroid Hormone. Endocrinology, 142(8), 3414–3420. doi:10.1210/endo.142.8.8303
[4] BARBE, P., LARROUY, D., BOULANGER, C., CHEVILLOTTE, E., VIGUERIE, N., THALAMAS, C., … LANGIN, D. (2001). Triiodothyronine-mediated up-regulation of UCP2 and UCP3 mRNA expression in human skeletal muscle without coordinated induction of mitochondrial respiratory chain genes . The FASEB Journal, 15(1), 13–15. doi:10.1096/fj.00-0502fje
[5] Baumgartner, C., da Costa, B. R., Collet, T.-H., Feller, M., Floriani, C., Bauer, D. C., … Rodondi, N. (2017). Thyroid Function Within the Normal Range, Subclinical Hypothyroidism, and the Risk of Atrial Fibrillation. Circulation, 136(22), 2100–2116. doi:10.1161/circulationaha.117.028753


































.gif)











































