- Joined
- Apr 9, 2021
- Messages
- 1,724
Evidence of MENT Aromatization & Conclusions About its Estrogenic Potency

MENT, along with other C19 androgens & 19-norsteroids, was incubated with recombinant human CYP19 + P450 & an NADPH complex, cells were transfected with luciferase, a reporter gene that "lights up," the relative light units (RLUs) indicating transactivation potencies. [100].
MENT's aromatic product (7α-methylestradiol; 7α-ME) is more than 4X (!) as potent ("efficacious," a bad thing here) than estradiol (E2) in ER-containing cells. [100]. Efficacy is determined by measuring the effect, e.g., growth (here, in breast cancer cells). The EC₅₀ (EC50) is determined by the concentrations at which the ligand triggers growth (this may be confirmed by measurements of cell cycle progression (i.e., the S-phase entry during the cell cycle).
The binding affinity (IC₅₀) of MENT's aromatic product (7α-methylestradiol) is 102% that of estradiol [100], which is typically used as the reference compound for ER binding given its noteworthy efficacy, potency, and affinity for the ERα receptor, in the literature.
Given the findings of Attardi et al, in comparing the rate of aromatization between MENT and Nandrolone, that "At 180 min, about 23% of MENT was converted to 7α-ME and about 13% of 19-NT to E2," knowing that Nandrolone aromatizes at 20% the rate of T [101], we can deduce that MENT aromatizes at roughly 35% the rate of T... to 7α-ME (an aromatic product with four-fold E2's potency, i.e., for causing growth in breast cancer cells). Simple multiplication of the rate of aromatization (35%) * EC50(7α-methylestradiol) * RBA(7α-methylestradiol) ≈ a 40% greater growth potential in ER-containing cells than T.
Keep in mind that you also need to consider the rate of breakdown of the produced 7α-methylestradiol, as well as the estrogenic potency of the resulting metabolites (Peter Bond, Oct 20 2021).
This would seem to support the anecdotes that MENT is quite a potent gynecomastic agent. With consideration of practical use, if the MENT dosage is ~70% the Test dosage it's on par with the estrogenicity of T, and given the aromatization to 7α-ME rather than E2, this consequence is unlikely to be reflected in bloodwork results. By way of comparison, e.g.: 35 mg daily of MENT E ≈ as estrogenic as 350 mg of Test E weekly.
References
[100] Attardi BJ, Pham TC, Radler LC, Burgenson J, Hild SA, Reel JR. Dimethandrolone (7alpha,11beta-dimethyl-19-nortestosterone) and 11beta-methyl-19-nortestosterone are not converted to aromatic A-ring products in the presence of recombinant human aromatase. J Steroid Biochem Mol Biol. 2008;110(3-5):214-222. doi:10.1016/j.jsbmb.2007.11.009
[101] Ryan, Kenneth J. “Biological aromatization of steroids.” Journal of Biological Chemistry 234.2 (1959): 268-272.
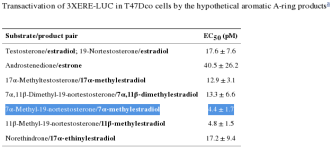
MENT, along with other C19 androgens & 19-norsteroids, was incubated with recombinant human CYP19 + P450 & an NADPH complex, cells were transfected with luciferase, a reporter gene that "lights up," the relative light units (RLUs) indicating transactivation potencies. [100].
MENT's aromatic product (7α-methylestradiol; 7α-ME) is more than 4X (!) as potent ("efficacious," a bad thing here) than estradiol (E2) in ER-containing cells. [100]. Efficacy is determined by measuring the effect, e.g., growth (here, in breast cancer cells). The EC₅₀ (EC50) is determined by the concentrations at which the ligand triggers growth (this may be confirmed by measurements of cell cycle progression (i.e., the S-phase entry during the cell cycle).
The binding affinity (IC₅₀) of MENT's aromatic product (7α-methylestradiol) is 102% that of estradiol [100], which is typically used as the reference compound for ER binding given its noteworthy efficacy, potency, and affinity for the ERα receptor, in the literature.
Given the findings of Attardi et al, in comparing the rate of aromatization between MENT and Nandrolone, that "At 180 min, about 23% of MENT was converted to 7α-ME and about 13% of 19-NT to E2," knowing that Nandrolone aromatizes at 20% the rate of T [101], we can deduce that MENT aromatizes at roughly 35% the rate of T... to 7α-ME (an aromatic product with four-fold E2's potency, i.e., for causing growth in breast cancer cells). Simple multiplication of the rate of aromatization (35%) * EC50(7α-methylestradiol) * RBA(7α-methylestradiol) ≈ a 40% greater growth potential in ER-containing cells than T.
Keep in mind that you also need to consider the rate of breakdown of the produced 7α-methylestradiol, as well as the estrogenic potency of the resulting metabolites (Peter Bond, Oct 20 2021).
This would seem to support the anecdotes that MENT is quite a potent gynecomastic agent. With consideration of practical use, if the MENT dosage is ~70% the Test dosage it's on par with the estrogenicity of T, and given the aromatization to 7α-ME rather than E2, this consequence is unlikely to be reflected in bloodwork results. By way of comparison, e.g.: 35 mg daily of MENT E ≈ as estrogenic as 350 mg of Test E weekly.
References
[100] Attardi BJ, Pham TC, Radler LC, Burgenson J, Hild SA, Reel JR. Dimethandrolone (7alpha,11beta-dimethyl-19-nortestosterone) and 11beta-methyl-19-nortestosterone are not converted to aromatic A-ring products in the presence of recombinant human aromatase. J Steroid Biochem Mol Biol. 2008;110(3-5):214-222. doi:10.1016/j.jsbmb.2007.11.009
[101] Ryan, Kenneth J. “Biological aromatization of steroids.” Journal of Biological Chemistry 234.2 (1959): 268-272.


































.gif)












































