I KNEW THERE WAS A REASON I LOVED TEST ISO !!
A few Things about Esters that I wasn’t quite aware of and neither were most of these multiple studies was done 2020-2022 and out online later :
Lab Science - they tested no ester base, acetate, propionate, phenylpropionate, and Isocaproate (Enanthate was used as a study market with a lot of data
The Results :
(TPro, TIso) pairs show a better match out of all the esters! Test ace and Test Iso have the highest Coulombic energy(scientifically) or crystal energy(so a good blend as well if you are a beast in that

lab. So the blend of Prop/Iso is superior (to the scientists).
(More science but look at the * part)
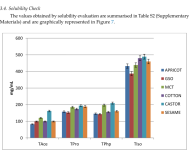
Depending on the length of the ester, it is observed that the shortest ester (the acetate)and has the lowest solubility, while the longest ester (isocaproate) while the longest ester (isocaproate) has a roughly

*four-fold greater greater solubility*. Propionate and phenylpropionate esters have similar solubility and slightly higher values than acetate. Phenylpropionate has six more carbon atoms than than acetate. Although phenylpropionate has six more carbon atoms than propionate, the propionate solubilities are similar, so it can be noted that is what matters. About increasing solubility (the phenyl ring does not lead to an increase in solubility). It is worth mentioning that *solubility correlates with the half-life* of the prodrug, so acetate, which has the shortest half-life, has the lowest solubility. Test isocaproate, which has the longest half-life, has the highest solubility. Out of the six mixtures analysed, it is observed that castor oil can support the highest solubility without crashing (crystallisation of compound) and to a lesser extent MCT (for solubility without crashing (crystallisation of compound) and to a lesser extent MCT (for TAce, TPro, and TPhp), at the same time behaving as solvents as well. This is interesting (TAce, TPro, and TPhp), at the same time behaving as solvents as well because MCT is characterised by the lowest value of the viscosity coefficient, while castor is the most viscous so it holds.
View attachment 171963

The solubility of the four derivatives was tested to evaluate the changes based on the added ester functionalities, and it was found that the shortest ester (acetate) has the lowest solubility, while the longest ester (isocaproate) is roughly four times greater; meanwhile, propionate and phenylpropionate are between the two and show similar values.
Just a recent research paper. But I’ve used iso (actually test iso/Tren hex ) and always like iso by itself as well. Apparently Test p/test iso would be a good combo . I realize Sustanon has 4 but low 30 prop /60iso . I’d love to bust out 100prop/150iso blend




































.gif)